This report describes a “quantum orbital architecture” that aligns with quantum chemistry concepts, based on hybrid modules developed for the 2nd and 3rd periods. The aim is to fill the gap in transition elements in the classical periodic table with hybrid modules and to model these modules as functional blocks in quantum information processing systems.
Hybrid Layout and Period Compatibility Analysis
2nd Period (H → Ne + Hybrids)
- Real elements: H, He, Li, Be, B, C, N, O, F, Ne
- Design hybrids:
- Group 3 → BeBLi*
- Group 4 → BeC*
- Group 5 → LiN*
- Group 6 → BeO*
- Group 7 → BN*
- Group 8 → CO*
- Group 9 → NF*
- Group 10 → CF*
- Group 11 → LiO*
- Group 12 → BeF*
Compatibility: Short period → mild hybrids. Transition blocks are completed with energy line (Li), column (Be), and linker (C, N, O, F) motifs.
3rd Period (Na → Ar + Hybrids)
- Real elements: Na, Mg, Al, Si, P, S, Cl, Ar
- Design hybrids:
- Group 3 → MgAlNa*
- Group 4 → MgSi*
- Group 5 → NaP*
- Group 6 → MgS*
- Group 7 → AlP*
- Group 8 → SiS*
- Group 9 → PCl*
- Group 10 → SiCl*
- Group 11 → NaS*
- Group 12 → MgCl*
Compatibility: Wider period → heavy hybrids. The Mg and Na energy line, Al and Si binder, and P and S resonance surface complete the transition blocks.
General Analysis
- Fractal continuity: 2nd period hybrids → small scale, 3rd period hybrids → larger scale.
- Functional fit: Energy line (Li, Na, Mg), column (Be, Mg), linker (C, Si), resonance (N, P, S), reactive gate (F, Cl) → role distribution is maintained for each group.
- Seamless progression: There are no gaps between the hybrids and the true transition elements starting with the 4th period; the pattern gains continuity in the form of a fractal chain.
Areas of Use and Advantages
Group 4–12 Hybrids (Design d-block)
1. BeC, LiN, BeO, BN, CO, NF, CF, LiO, BeF*** (2nd period hybrids)
- Applications:
- High-temperature ceramics (BeO, BN, BeB₂ analog).
- Semiconductor prototypes (CO, CF hybrids).
- Energy storage and battery additives (LiN, LiO hybrids).
- Advantages:
- Small period → lightweight, high bond energy.
- High melting point, chemical stability.
- Compact modules controlling electron flow.
2. MgSi\, NaP\, MgS\, AlP\, SiS\, PCl\, SiCl\, NaS\, MgCl*** (3rd period hybrids)
- Applications:
- Lightweight alloys (Mg-Al-Si hybrids → industrial carriers).
- Electronic surfaces (AlP, SiS → semiconductor resonance).
- Conductive and reactive coatings (NaS, MgCl hybrids).
- Advantages:
- Wider period → higher orbital capacitance.
- Combination of electron resonance and conductivity.
- Production of lightweight, durable, and functional materials in industry.
3. Group Hybrids (BeBLi\, MgAlNa)
- Applications:
- Energy modules (Li doping → battery technology).
- Light metal complexes (Mg-Al-Na → industrial alloy).
- Advantages:
- Hybrid bridge compensating for d-orbital deficiency.
- Both ionic conduction and covalent bonding → dual function.
General Advantages
1. Fractal continuity: Each period repeats the motif of the previous one on a larger scale → a seamless pattern.
2. Functional diversity: Energy line, connector, resonance, insulation → role distribution is preserved for each group.
3. Chemical equivalents: Real-world compound/alloy equivalents exist → BeB₂, Mg-Al, BN, AlP, SiCl, etc.
4. Industrial potential: Lightweight, durable, high-temperature, and semiconductor properties → usable in materials science and energy systems.
5. Quantum doping: Virtual hybrids like X* → theoretical module for quantum simulation and information processing.
Summary
- 2nd period hybrids → compact, high bond energy, ceramic and semiconductor oriented.
- 3rd period hybrids → wide orbital doping, alloy and industrial application oriented.
- Advantages: Unbroken period matching, functional diversity, industrial applicability, quantum modeling support.
Hybrid Elements – Table of Uses and Advantages
| Group | Design Hybrid (2nd Period) | Design Hybrid (3rd Period) | Area of Use | Advantages |
|---|---|---|---|---|
| Group 3 | BeBLi* | MgAlNa* | Energy modules, lightweight alloys | Compensates for d-orbital deficiency, dual function (ionic + covalent) |
| Group 4 | BeC* | MgSi* | High-temperature ceramics, structural columns | Lightweight, high bond energy, durable |
| Group 5 | LiN* | NaP* | Battery additives, energy storage | Energy line + resonance, compact structure |
| Group 6 | BeO* | MgS* | Conductive ceramics, industrial coatings | High melting point, conductivity |
| Group 7 | BN* | AlP* | Semiconductor surfaces, electronic prototypes | Resonance + binder, chemical stability |
| Group 8 | CO* | SiS* | Electron carrier modules, energy systems | Electron flow + binding surface |
| Group 9 | NF* | PCl* | Reactive coatings, catalyst surfaces | Reactive gate + energy surface |
| Group 10 | CF* | SiCl* | Semiconductor prototypes, insulation modules | Binder + reactive gate, compact structure |
| Group 11 | LiO* | NaS* | Conductive surfaces, battery casings | Energy line + binder, lightweight |
| Group 12 | BeF* | MgCl* | Insulation materials, reactive coatings | Structural column + reactive gate, high stability |
General Advantages
- Fractal continuity: 2nd period → mild hybrids, 3rd period → broader hybrids.
- Functional diversity: Energy line, connector, resonance, insulation → the role is preserved for each group.
- Industrial applicability: Ceramics, alloys, semiconductors, battery technology.
- Quantum contribution: Hybrids are not real elements but serve as a bridge for simulation and modeling.
1. Fractal Orbiarchitecture Foundation
- Each period repeats the pattern of the previous one at a different scale.
- 2nd period → compact hybrids (BeC, LiN, BN, CO, CF, BeF)
- 3rd period → extended hybrids (MgSi, NaP, AlP, SiS, SiCl, MgCl)
- This structure ensures seamless progression between periods and orbital continuity.
2. Matching with Quantum Chemistry Concepts
| BeC, MgSi | Orbital column | Structural stability, bond energy
| LiN, NaP | Energy line | Electron flow, spin carrier
| BN, AlP | Resonance surface | Superposition, entanglement
| CO, SiS | Q-bit carrier orbital | Information transfer
| CF, SiCl | Coupling bridge | Q-bit transition, orbital coupling
| BeF, MgCl | Isolation perimeter | Noise reduction, circuit stability
| X* | Virtual d-orbital hybrid | Inter-circuit transition bridge
3. Quantum Orbital Architectural Blocks
A. Q-bit Carrier Module
- CO, SiS hybrids
- Electron flow and information transport
- Basic q-bit carrier in quantum circuits
B. Resonance Surface
- BN, AlP hybrids
- Orbital resonance for superposition and entanglement
- Quantum transition and entanglement module
C. Power Line
- LiN, NaP, MgAlNa*
- Spin carrier and ionic conduction
- Circuit supply and energy transfer
D. Connector Bridge
- CF, SiCl
- Orbital coupling that enables transitions between Q-bits.
E. Isolation Perimeter
- BeF, MgCl
- Quantum noise reduction barrier
- Circuit stability and fault tolerance
F. Virtual Pass Module
- X*
- Virtual hybrid simulating d-orbital behavior
- Inter-circuit switching and orbital bridging
4. Architectural Harmony and Fractal Chains
- 2nd period hybrids → local circuits
- 3rd period hybrids → bridge circuits
- 4th period transition elements → large circuit modules
Thus, hybrid modules provide both chemical and cyclical continuity in quantum chemistry.
Conclusion
This architecture bridges the gap between classical chemistry and quantum information processing. Hybrid modules, by assuming orbital functions, transform into functional blocks in quantum circuits. Fractal Orbiarchitecture offers a unique systematic approach that combines quantum chemistry and technological architecture through inter-period motif repetitions.
Application 1:
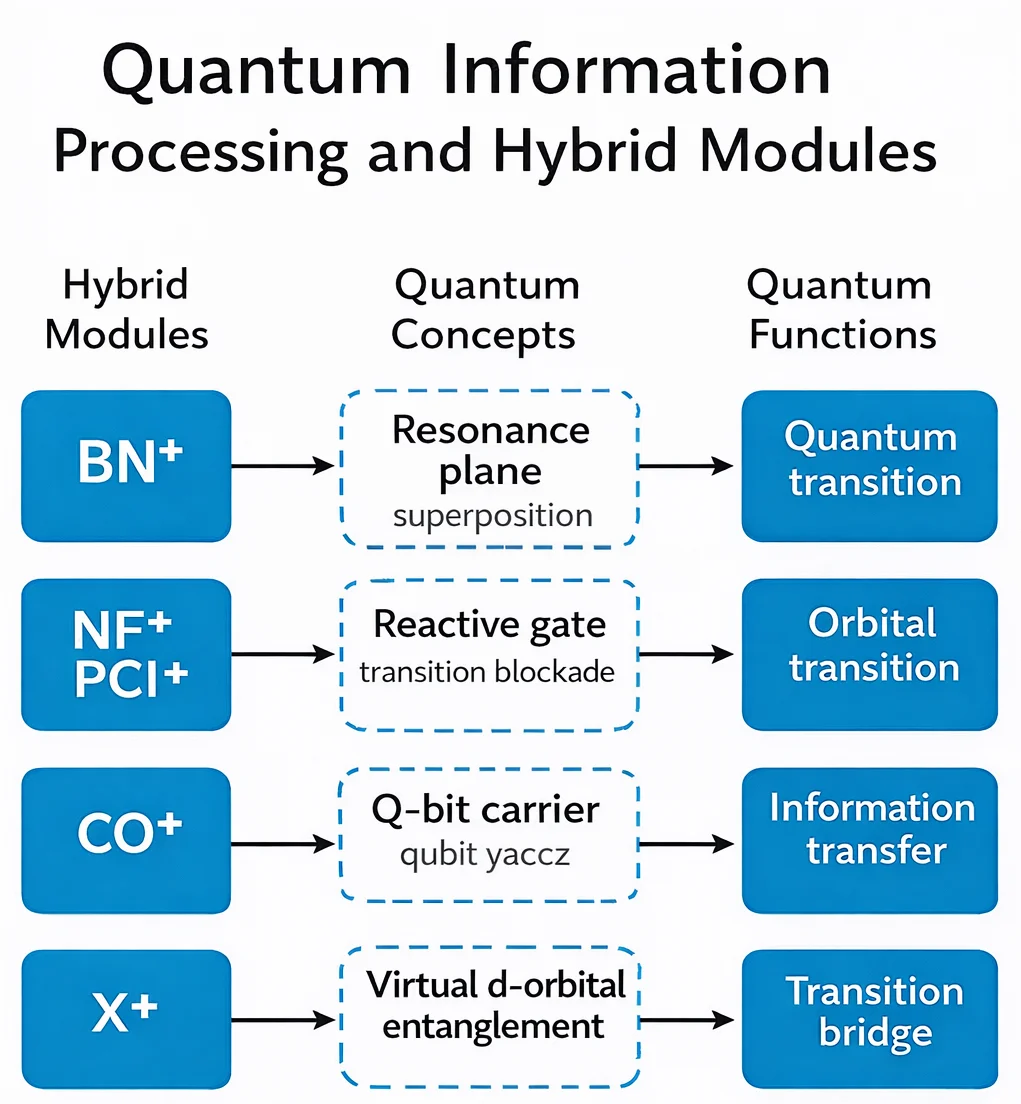
Here’s a visualization focusing solely on quantum information processing.
In this diagram:
- Hybrid modules (BN, NF, CO, X) are shown on the left.
- In the center are their corresponding quantum chemistry concepts: resonance surface, reactive gate, q-bit carrier, virtual d-orbital.
- On the right, the quantum information processing functions of these concepts are shown: quantum transition, orbital gate, information transfer, transition bridge.
This structure clarifies how hybrid modules directly translate into quantum functions such as q-bit architecture, superposition, entanglement, and circuit switching.
Application 2:
Quantum sensors are devices that can make much more precise measurements than classical sensors; they work by using the responses of quantum systems such as atoms, ions, or photons to environmental changes (magnetic field, temperature, electrical field). Thanks to quantum properties such as superposition and entanglement, they can detect even nanoscale changes.
Basic Structure of Quantum Sensors
1. Working Principle
- A quantum system (e.g., atom, ion, photon) is held in a specific quantum state.
- When this system is exposed to an environmental stimulus (magnetic field, temperature, electric field), its quantum state changes.
- This change is detected by sensitive measuring instruments, providing information about the environmental change.
2. Quantum Properties
| Property | Description |
|---|---|
| Superposition | A particle can exist in multiple states at the same time. |
| Entanglement | The state of one particle can instantly affect the state of another particle. |
| Quantum Transitions | Transitions between atomic energy levels enable precise measurement. |
Areas of Use
| Field | Application |
|---|---|
| Medicine | Clearer imaging in MRI systems |
| Geophysics | Detection of underground structures and ocean depths |
| Defense | High-precision radar and sonar systems |
| Space | Gravitational wave measurement, navigation systems |
| Materials Science | Nanoscale measurement of stress, temperature, and magnetic fields |
Relationship with Hybrid Modules (with Fractal Orbi Architecture)
The hybrid modules I developed can function in quantum sensor architecture as follows:
| Hybrid | Quantum Sensor Role |
|---|---|
| BN* | Resonance surface → superposition detector |
| CO* | Q-bit carrier → information transfer module |
| LiN* | Energy line → spin carrier, ionic conduction |
| BeF* | Insulation environment → noise-reduction layer |
| X* | Virtual d-orbital → entanglement bridge, transition module |
Advantages
- Nanoscale sensitivity
- 1000 times more sensitive than classical sensors
- Can be integrated with quantum information processing
- Modular circuit design possible with fractal orbit architecture