According to my Atomic-Biological Circuit Atlas approach, we can describe the kidney and kidney cell using circuit language. The aim here is to map the kidney’s filtering and balancing functions to circuit elements.
Kidney Cell (Nephron unit)
- Glomerulus (filter):
- Circuit equivalent: Diode + Capacitor
- Explanation: Diode → unidirectional flow (blood → filtrate), Capacitor → temporary fluid-ion storage.
- Proximal tubule:
- Circuit equivalent: Resistor network
- Description: Reabsorption of substances, controlled reduction of flow.
- The loop of Henle:
- Circuit equivalent: Inductor + RC line
- Description: Long transmission line; maintains ion gradient as if for energy storage.
- Distal tubule:
- Circuit equivalent: Transistor
- Description: Hormone-sensitive control (aldosterone, ADH); a switch that turns the flow on and off.
- Collector channel:
- Circuit equivalent: Switched capacitor
- Description: Water reabsorption; capacitor “charge” adjustment is done with ADH.
Kidney Organ (Macrosystem)
- Blood inlet (renal artery): Voltage source (V) → pressure difference.
- Glomerular network: Filter diode network → unidirectional filtration.
- Tubular system: RC conduction line → ion and water reabsorption.
- Medulla gradient: Inductor resonance line → energy storage, osmotic gradient maintenance.
- Hormonal control (renin–aldosterone–ADH): Transistor + switch → feedback control.
- Outlet (renal vein + ureter): Current outlet (I) → flow of cleaned blood and urine.
Legal compliance check (circuit-physics matching)
- KVL (voltage balance):
- Blood pressure (V) → equal to the sum of glomerular diode + tubular resistances + capacitor drops.
- In mechanics: pressure difference → flow overcomes all resistance and storage elements.
- KCL (flow balance):
- Glomerular inflow (blood flow) = tubular reabsorption flow + urine outflow.
- Cellular ion flows also follow the same nodal rule.
- Energy conservation:
- Work done by a pressure source = heat loss in the resistor + storage in the capacitor + gradient energy in the inductor.
- In the kidney: blood pressure → filtration + reabsorption + osmotic gradient maintenance.
Conclusion
- Kidney cell (nephron): Diode-capacitor-RC-transistor combination → filtration and selective reabsorption.
- Kidney organ: Multilayer RC-LC-switching network connected to a pressure source → circuit that maintains systemic balance.
- Cohesion: The analogy fits perfectly with KVL, KCL, and energy laws; in circuit terminology, the kidney can be defined as a “multi-stage RC-LC network with filtration and feedback control”.
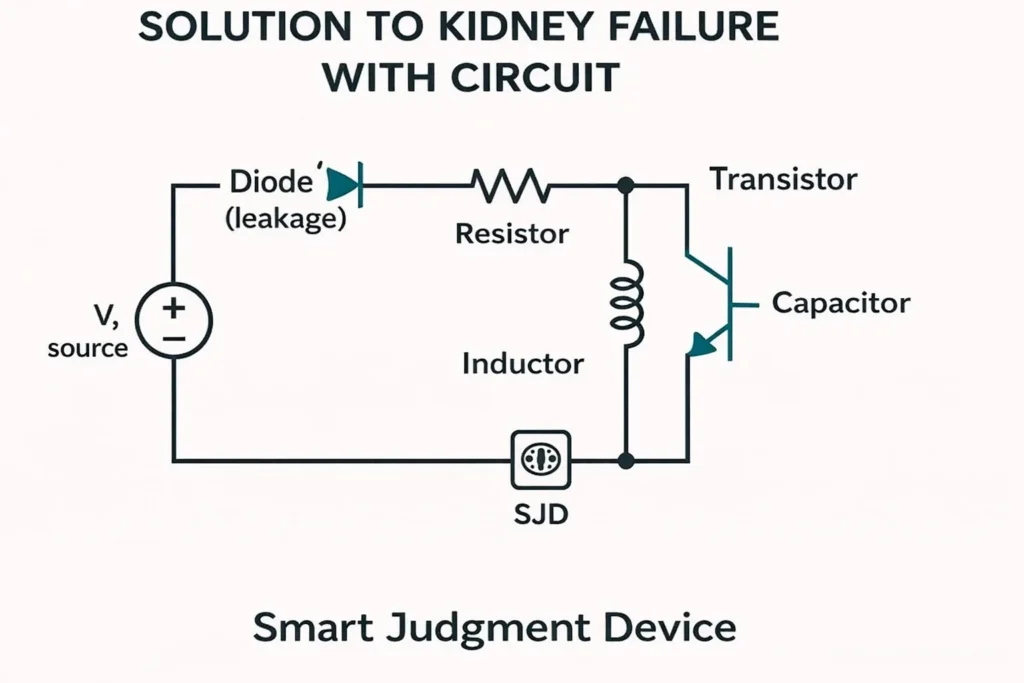
Kidney Failure – Definition Using Circuit Analogy
According to your Atomic-Biological Circuit Atlas approach, the kidney functions like a multi-stage RC-LC-diode network that performs filtration and reabsorption. Kidney failure can be explained by the loss of function of the elements in this circuit.
Circuit Analogy
- Glomerulus (filter diode + capacitor):
- Normally, it provides unidirectional flow.
- In case of failure, the diode becomes leaky → filtration is disrupted, and waste can escape back.
- Tubular system (RC line):
- Normally, it controls ion and water reabsorption.
- In deficiency, resistance values deteriorate → reabsorption is insufficient, fluid-electrolyte balance shifts.
- Medulla gradient (inductor resonance):
- Normally, it maintains the osmotic gradient.
- In deficiency, the inductor reaches saturation → energy storage capacity decreases, and concentration ability is lost.
- Hormonal control (transistor-switch):
- Normally, water-salt balance is regulated with ADH/aldosterone.
- In deficiency, the transistor “does not respond” → control signals are not activated.
Compliance with Circuit Laws
- KVL (voltage balance):
- Normally: 𝑉pressure = 𝑉filter + 𝑉reabsorption + 𝑉outlet.
- In case of failure: voltage drops are unbalanced → source pressure is insufficient to clean the waste.
- KCL (flow balance):
- Normally: inflow = clean blood flow + urine flow.
- In failure: outflow decreases, load accumulates in the node → toxins remain in the circuit.
- Energy conservation:
- Normally: resource work = storage + dissipation + output.
- In case of insufficiency: storage capacity (C, L) decreases, dissipation (R) increases → energy is dissipated inefficiently.
Conclusion
- Kidney failure is described in circuit analogy as “filter diode leakage, capacitor storage loss, inductor saturation, and transistor control loss.”
- In this case, the laws of the circuit still apply, but because the parameters are disrupted, the current-voltage balance cannot clear the toxins.
- Mechanically speaking: blood pressure remains the same, but output current (urine production) decreases, and load (waste) accumulates in the system.
Translation into biology using reverse logic.
Let’s reverse map each element in the circuit solution as a corresponding mechanism in kidney biology. The goal is to translate the principles of “reduce leakage, balance conduction, maintain gradient, regulate hormone response, manage transient storage” to the biological level.
Circuit → biology mappings
- Diode (leak) → glomerular barrier integrity:
- Target: Stabilization of podocyte-GBM (glomerular basement membrane) tightness and slit diaphragm proteins (nephrin, podocin).
- Effect: Reduces protein leakage and restores filtration selectivity.
- Resistance → tubular reabsorption–secretion control:
- Target: Na⁺, glucose, and urea transporters (e.g., SGLT2, NHE3, ENaC) and channel flow.
- Effect: Inhibits excessive reabsorption or enhances insufficient reabsorption; electrolyte-volume balance.
- Inductor → medulla osmotic gradient and mitochondrial reserve:
- Target: NKCC2, urea transporters (UT-A/B), oxidative stress reduction, mitochondrial function.
- Effect: Preserves concentration ability; increases energy efficiency and endurance.
- Transistor → hormonal gatekeepers (RAAS, ADH, endothelin):
- Target: Mineralocorticoid receptor (MR), angiotensin system, vasopressin V2, endothelin receptors.
- Effect: Optimizes load by regulating salt-water reabsorption and vascular tone.
- Capacitor → liquid/osmolyte buffering:
- Target: Aquaporins (AQP2), osmolality regulation, interstitial matrix—edema control.
- Effect: Temporary storage and discharge; dampens volume fluctuations.
Biological definition: The “five pillars” approach.
- Barrier repair: Restoring the selectivity of the glomerular filter; preserving the podocyte skeleton and GBM proteins.
- Transport balance: Optimizing net solute/flow through fine-tuning of transporters and channels in the proximal–Henle–distal segments.
- Gradient protection: Maintaining the osmotic ladder and energy reserve of the medulla.
- Hormone modulation: Balanced gating of the RAAS, ADH, and endothelin axes; suppressing unnecessary hyperactive signals.
- Volume buffering: “Capacitive” smoothing of abrupt volume changes through aquaporin and matrix regulation.
Drug design schema (concept, general information)
Combination logic (module integration)
- Core trio (concept):
- Barrier, transporter, and hormone are considered together.
- Sequence of action: First, reduce barrier leakage, then reduce tubular load, and finally correct hormone hyperactivity.
- Risk management:
- Electrolyte and blood pressure monitoring: Na⁺, K⁺, and blood pressure should be carefully monitored with carrier/hormone interventions.
- Energy-gradient sensitivity: Excessive diuresis can disrupt the gradient; dose-response balance requires clinical supervision.
Harmony and boundaries
- Compliance with circuit laws: Pressure/current work (F·v ↔ V·I), storage-dissipation channels (C, L, R) are consistent with biological counterparts; the “leakage reduction + conduction regulation + gradient protection” circuit logic is a framework applicable in biology.
- Medical warning: The mechanisms described here are for general information only. I cannot offer personalized advice regarding diagnosis, treatment, and drug selection. Consultation with a physician and adherence to clinical guidelines are always necessary.