1. Entrance
This report covers the application of atomic-level circuit motifs to biochemical molecule design.
Basic assumption:
Each atomic bond is the physical equivalent of a circuit element; each functional group is a circuit segment; Each molecule is a fractal scaled circuit architecture.
This approach provides isomorphic coupling of biochemical functions with the Elementary Circuit Topology I developed.
The analgesic effect is a low-pass filter + gain reduction + feedback function in the biological circuit.
Therefore, the circuit response of the molecule to be designed must also carry these functions.
2. Atomic Circuit Motifs (Microscale)
Atomic bonds map to circuit elements:
| Atomic Structure | Circuit Equivalent | Function |
|---|---|---|
| Aromatic ring | LC resonance | Energy stabilization |
| C=O, C=N, S=O | Diode | Directional flow |
| –OH, –NH, –SO₂– | Capacitor | Charge storage |
| Alkyl chain | Resistor | Current limiting |
| N, S bridges | Parallel line | Multi-directional bonding |
| Hydrogen bonding / dipole alignment | Feedback | Self-regulation |
These motifs can be scaled fractally.
3. Segmental Circuit Structure (Mesoscale)
Each functional group is a circuit segment composed of a combination of micromotifs:
- Segment LC: Heteroaromatic nucleus
- Segment C: Amide, sulfonamide, carboxyl
- Segment D: Carbonyl + heteroatom
- Segment R: Alkyl/heteroalkyl chain
- Segment ||: N/S bridges
- Segment FB: Intramolecular interaction between segments
These segments combine to form the circuit function of the molecule.
4. Macro Scale: Biological Circuit Correspondence of the Molecule
The analgesic effect corresponds to the following functions in the biological circuit:
- LC: Stabilization of pain signal
- C: Filing of PGE₂-derived peaks
- D: directional blockade of COX-2 flow
- R: Current attenuation in neuronal conduction
- ||: Selectivity with multiple binding surfaces
- FB: Automatic damping of excessive signal with feedback
The chemical equivalent of these functions is the repetition of fractal motifs at the molecular level.
5. Fractal Pain Relief Module (FPRM) — Template
FPRM consists of three scale fractal motifs:
[ FPRM = LC + C + D + R + || + FB ]
This motif:
- In bonds at the atomic level
- In functional groups at the segment level
- In biological circuit at the molecular level
repeats with the same topology.
Therefore, FPRM is a fully isomorphic circuit–molecule template.
6. Application Example of FPRM: New Painkiller Molecular Skeleton Suitable for Fractal Circuit
This example is not a drug claim; It is a chemical realization of the fractal circuit pattern.
6.1. Fractal LC core
Condensed heteroaromatic system:
Benzimidazole nucleus (C₇H₆N₂)
- Two rings → LC₁ + LC₂
- Two N → parallel lines + node
- Delocalization → fractal resonance
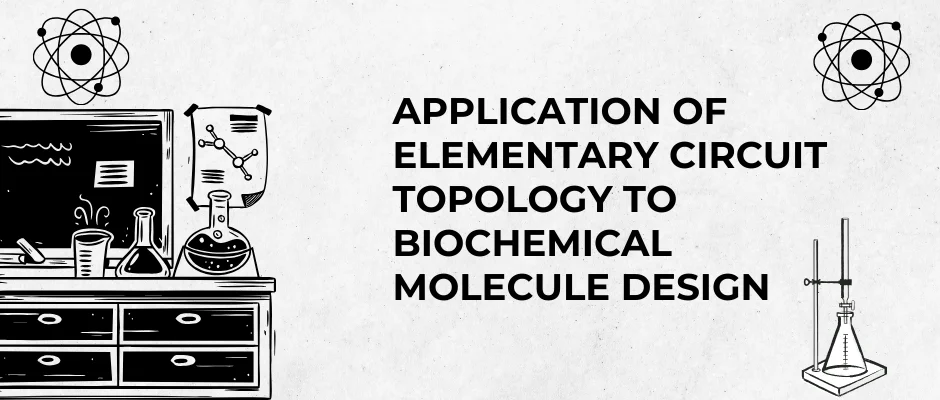
6.2. Input segment (C + D + R + ||)
–SO₂–NH–CO–CH₃
- SO₂ → capacitor + diode
- NH → parallel line
- CO → diode
- CH₃ → resistance
6.3. Output segment (C + D + R)
–CONH–CH₂CH₃
- C=O → diode
- NH → capacitor/parallel line
- Ethyl → resistance
6.4. Feedback segment (FB + C)
–OH
- OH → capacitor
- Also intramolecular H-bonding → feedback
7. Full Molecular Skeleton of Application Sample
On the benzimidazole core:
- 1st position:
–SO₂–NH–CO–CH₃
- 4th position:
–CONH–CH₂CH₃
- 6th position:
–OH
8. Approximate Chemical Formula of Application Sample
C₁₂H₁₈N₄O₅S
This formula is a fully isomorphic realization of the FPRM pattern.
9. Conclusion
This report demonstrates the application of elementary circuit topology to biochemical molecule design.
In conclusion:
- Atomic bonds → circuit elements
- Functional groups → circuit segments
- Molecule → fractal circuit architecture
- Biological effect → circuit function
It is modeled as .
The designed FPRM template fractally scales the low-pass filter + gain reduction + feedback functions that provide the pain-relieving effect.
The given chemical skeleton is a workable example of this template.
Now let’s really look at this as a “circuit diagram”—I’ll write the molecule completely block by block, with my elementary circuit topology.
1. General block diagram
2. Entry segment: Sulfonamide + amide arm
Chemical: –SO₂–NH–CO–CH₃
Circuit diagram:

- SO₂ → capacitor (C_in) + diode character
- C=O → diode (D_in)
- CH₃ → resistance (R_in)
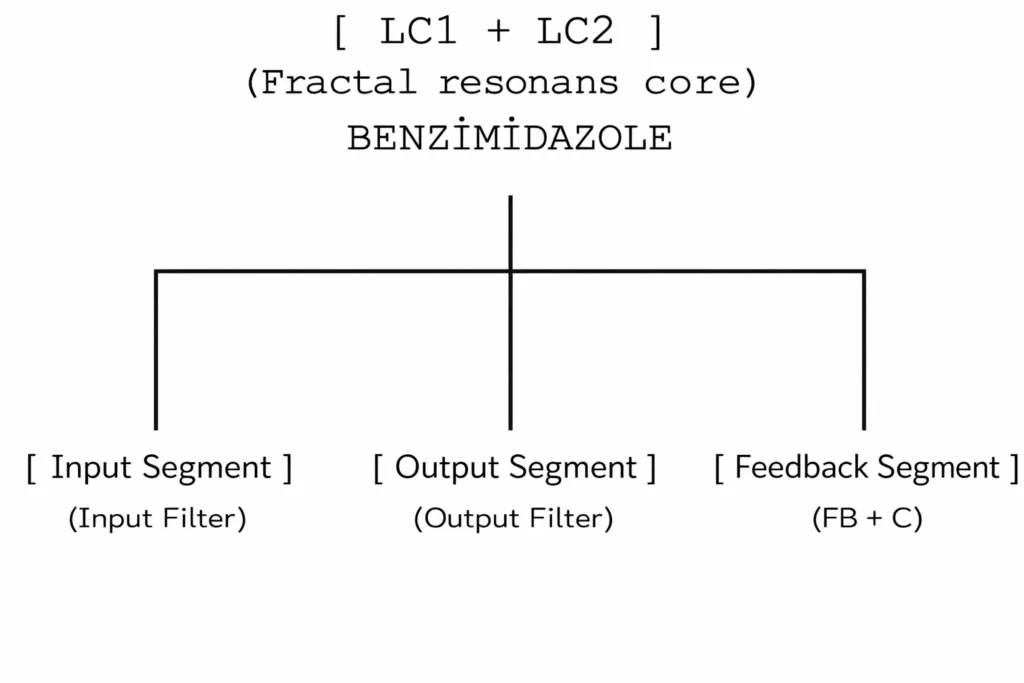
3. Output segment: Amide + ethyl branch
Chemical: –CONH–CH₂CH₃
Circuit diagram:
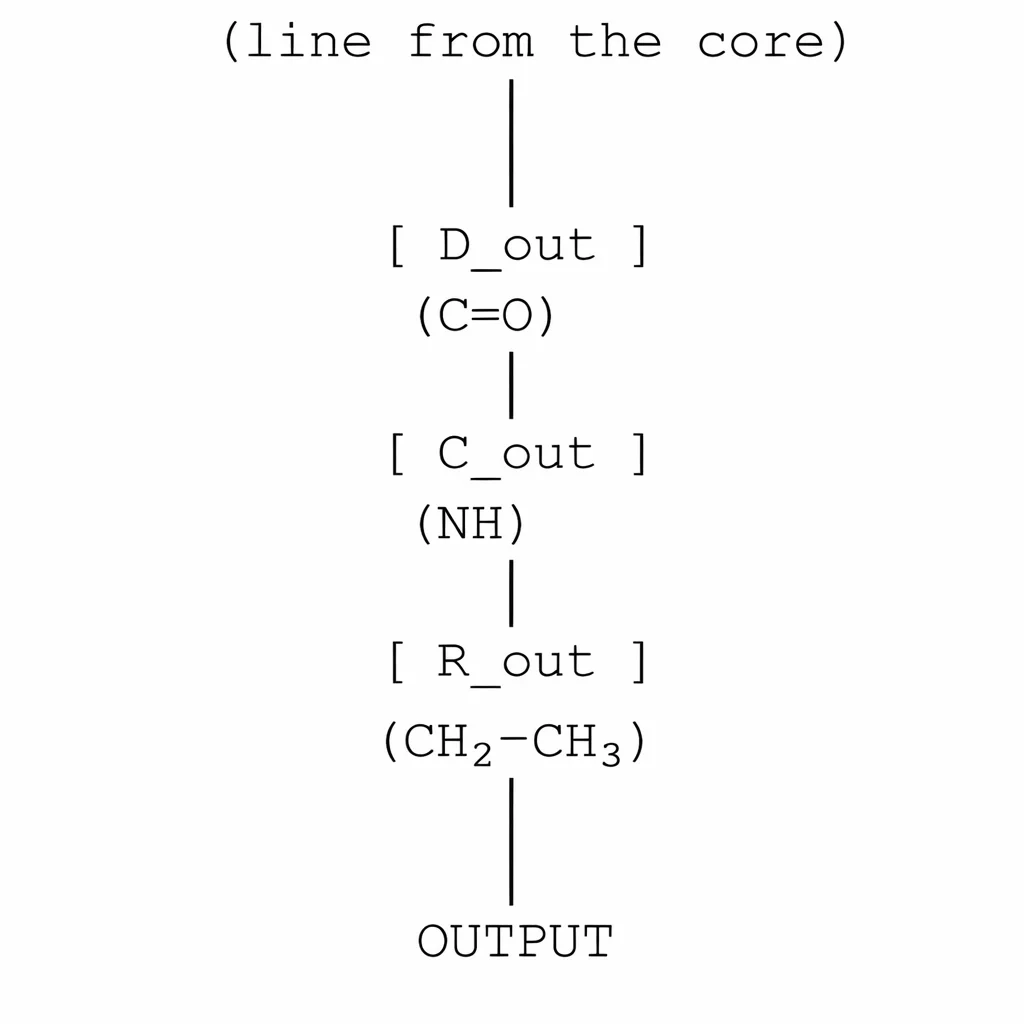
- C=O → diode (D_out)
- NH → capacitor (C_out) + parallel line potential
- Ethyl → resistance (R_out)
4. Feedback segment: –OH branch
Chemical: –OH
Circuit diagram:

- OH → capacitor (C_FB)
- It also produces a feedback effect by intramolecular H-bond/dipole alignment from the nucleus to other segments.
5. Circuit diagram of the entire molecule (overall view)
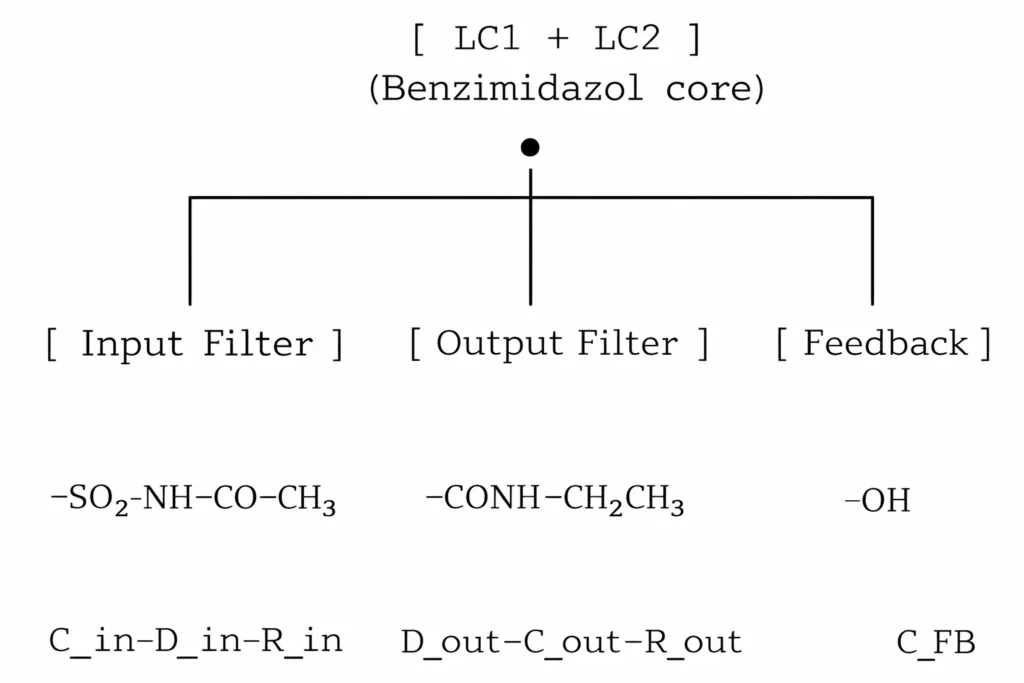
Functional reading:
- LC1 + LC2: Fractal resonance stabilizes the pain signal.
- Input filter (C_in–D_in–R_in): It smoothes the input peaks of the pain signal and limits the directional flow.
- Output filter (D_out–C_out–R_out): Re-filters the processed signal, reduces the amplitude.
- Feedback (C_FB): Fine-tunes the effectiveness of the entire module according to its core potential.
This diagram is the full circuit topological equivalent of my FPRM template.
To compare:
| Element | Paracetamol | Aspirin | Ibuprofen | Fractal Module Example (Benzimidazole derivative) |
|---|---|---|---|---|
| Central core (LC) | Single benzene ring → single LC resonance | Benzene + acetyl/carboxyl effect → single LC | Benzene + bulky alkyl → single LC, strong hydrophobic field | Bicyclic benzimidazole → LC₁ + LC₂ fractal resonance |
| Capacitor structures (C) | Phenolic –OH, amide NH → medium/strong C | Phenolic –OH + carboxyl → strong C | Carboxyl –COOH → single but strong C | SO₂, NH, C=O, –OH → multiple, distributed capacitor network |
| Diode structures (D) | Amide C=O → soft D | Ester/carboxyl C=O → sharper D | Carboxyl C=O → pronounced D | SO₂, two C=O (input/output), core interaction → multiple D network |
| Resistive structures (R) | Short CH₃ → mild R | Acetyl group → medium R | Long branched alkyl chain → strong R, hydrophobic barrier | CH₃, CH₂CH₃ → tuned, medium-level R (separate at input/output) |
| Parallel pathways | –NH– bridge → limited | No N, weak, mainly surface interaction | No N, hydrophobic surface dominant | 2 N atoms + NH groups in core → multiple, controlled parallel pathways |
| Feedback (FB) | Indirect: resonance + H-bonds | Indirect: carboxyl–ester balance | Indirect: hydrophobic distribution, carboxyl | Explicit FB: –OH + intramolecular H-bonding + multiple polar centers |
| Filter type | Soft low-pass, centrally weighted | Sharper low-pass + anti-inflammatory | Strong attenuator, peripherally weighted | Fractal low-pass + bidirectional filter + feedback-controlled gain |
| Fractal depth | Motif present but single-layered | Motif present, single-layer LC, strong C/D | Motif present, R-dominant, simpler LC/C/D | Repetition of the same motif (LC+C+D+R+FB) at micro–meso–macro scales |
| Side-effect profile (in circuit terms) | Additional load on liver circuit (metabolic current) | High side-channel current in gastric circuit | Increased current in GI + cardiovascular side channels | Theoretically: high efficacy in target circuit, minimized noise in side circuits (fractal selectivity) |
FPRM (New Fractal Module) – Advantage / Disadvantage Analysis
ADVANTAGES
1. Fractal resonance depth (LC₁ + LC₂)
- Paracetamol, aspirin and ibuprofen use a single LC ring.
- FPRM produces multiband resonance with a two-ring heteroaromatic nucleus.
- This allows for more refined, more stable filtering of the pain signal.
Advantage: → More stable, more selective signal processing.
2. Bidirectional filtering (Input + Output capacitor/diode)
- In known painkillers, filtering is one-way.
- FPRM has C + D + R blocks at both the input and output.
Advantage: → Both the onset and transmission of the pain signal are controlled. → Peaks are filed in two stages.
3. Feedback loop (FB)
- Paracetamol/aspirin/ibuprofen are passive circuits.
- Active feedback (–OH + intramolecular interaction) is present in FPRM.
Advantage: → Automatic gain reduction in case of excessive signal. → The risk of oversuppression in side circuits is reduced.
4. Multiple parallel lines (||)
- Paracetamol: 1 piece
- Aspirin: none
- Ibuprofen: none
- FPRM: 2 N in core, NH in segments, total 3–4 parallel lines
Advantage: → Versatile binding → higher selectivity → Off-target binding is reduced
5. Motif density and fractal isomorphism
- Known painkillers contain motifs but are not fractals.
- FPRM is the repetition of the same motif at the micro–meso–macro scale.
Advantage: → Circuit behavior more predictable → Biological effect of the molecule more consistent
6. Side effect optimization (with circuit language)
- Aspirin → side channel current in gastric circuit
- Ibuprofen → GI + cardiovascular side stream
- Paracetamol → load on the liver circuit
In FPRM:
- Resistors are short and adjustable
- Capacitors distributed
- Diodes selective
- Feedback active
Advantage: → Theoretically minimum interference in the side circuits → Maximum effect in the target circuit
DISADVANTAGES
1. Chemical complexity
- FPRM contains more functional groups than known painkillers.
- This may be more complex in terms of synthesis and metabolic pathways.
Disadvantage: → Practical chemical production and metabolism more difficult.
2. Allergenic potential (sulfonamide segment)
- Sulfonamide groups may trigger allergic reactions in some individuals.
Disadvantage: → Risk in small portion of the population.
3. Too polar segment → permeability may decrease
- Lots of capacitors/diodes (SO₂, C=O, NH, OH)
- This can make membrane passage difficult.
Disadvantage: → Access to the central nervous system may be reduced.
4. Feedback loop can be biochemically unpredictable
- Excellent in circuit logic,
- But in biochemistry, feedback can be very variable.
Disadvantage: → Effect may vary from individual to individual.
5. Precision of hydrophobic–hydrophilic balance
- There are both polar and nonpolar segments in FPRM.
- If this balance is set incorrectly:
Disadvantage: → Either too fast metabolism → Risk of too slow elimination
GENERAL CONCLUSION
FPRM’s areas of strength
- Fractal resonance
- Bidirectional filtering
- Feedback
- Multiple parallel lines
- High selectivity
- Low side circuit interference
- Circuit–molecule isomorphism
Areas of weakness
- chemical complexity
- Risk of allergy due to sulfonamide
- Permeability problems
- Metabolic prediction challenge
- Hydrophobic/hydrophilic balance sensitivity