Cancer cells are abnormal cells that, unlike normal cells, divide uncontrollably, damage surrounding tissues, and can spread to other parts of the body (metastasis). They are formed as a result of genetic mutations and acquire characteristics such as evading the immune system, becoming immortal, and altering energy production.
Basic Characteristics of Cancer Cells
| Feature | Description |
| Uncontrolled cell division | Cell cycle regulators are disrupted; it multiplies continuously. It reproduces continuously. |
| Apoptosis resistance | It is resistant to programmed cell death mechanisms. |
| Ability to metastasize | It can spread to other tissues; it is transmitted via blood and lymph. |
| Angiogenesis | It stimulates new blood vessel formation, thereby increasing its nourishment. |
| Change in energy production | It utilizes glucose even in the absence of oxygen (Warburg effect). |
| Evasion of the immune system. | It reduces MHC molecules and suppresses the immune response. |
| Genetic instability | DNA repair mechanisms break down; mutations accumulate. |
How does it form?
- Genetic mutations: Oncogene activation (e.g., RAS), inactivation of tumor suppressor genes (e.g., TP53).
- Epigenetic changes: DNA methylation, histone modifications.
- Environmental factors: Smoking, radiation, viruses (HPV, EBV), chemicals.
- Chronic inflammation: Continuous cell damage and renewal cycles increase the risk of mutations.
Differences Between Cancer Cells and Normal Cells
Cancer Cell Using Circuit Analogy
According to the circuit-biology approach:
- Transistor remaining constantly on → loss of cell cycle control
- Diode directionality disruption → metastasis and reverse flow
- Capacitor overcharging → energy and signal accumulation
- Increased noise source → genetic instability and entropy
Conclusion
A cancer cell is a type of cell that has deviated from normal cell behavior due to genetic and epigenetic abnormalities, and can grow and spread uncontrollably. Both biologically and using a circuit analogy, it can be modeled as a state where the system’s “control, direction, energy, and signal” mechanisms are disrupted.
Drug Definition – General Framework
A drug is a chemical or biological agent used to prevent, treat, or alleviate symptoms of disease by acting on a specific target in biological systems. Drugs generally consist of an active ingredient, excipients, and formulation components.
Basic Characteristics of Medicines
- Active ingredient: The molecule that produces the main therapeutic effect (e.g., antibiotic, antihypertensive).
- Excipients: Additives that increase the solubility, absorption, or stability of the drug.
- Pharmacokinetics: The absorption, distribution, metabolism, and excretion of a drug in the body.
- Pharmacodynamics: The effect of a drug on a cell or receptor.
- Dosage form: Tablet, capsule, injection, cream, aerosol, etc.
Drugs Using Circuit Analogy
According to the circuit-biology approach, a drug is like an external signal that adjusts a circuit parameter:
- Resistance-modulating drugs: Modulate ion channels or transporters (e.g., diuretics).
- Capacitor-modulating drugs: Affect intracellular storage/energy systems (e.g., antioxidants).
- Transistor-modulating drugs: Turn hormone receptors or signaling pathways on and off (e.g., steroid antagonists).
- Inducer-boosting drugs: Enhance energy metabolism and mitochondrial function.
Sample Drug Groups
| Group | Mechanism of Effect | Example |
| Antibiotic | It inhibits bacterial cell wall/protein synthesis. | amoxicillin |
| Antihypertensive | Lowers blood pressure | ACE inhibitors |
| Anticancer | It stops cell division. | Chemotherapy agents |
| Antiviral | It prevents virus replication. | Oseltamivir |
| Anti-inflammatory | It reduces inflammation. | NSAIDs |
Notes
The information provided here is for general purposes only. The choice, dosage, and use of medication require individual medical advice; therefore, a physician or pharmacist should always be consulted.
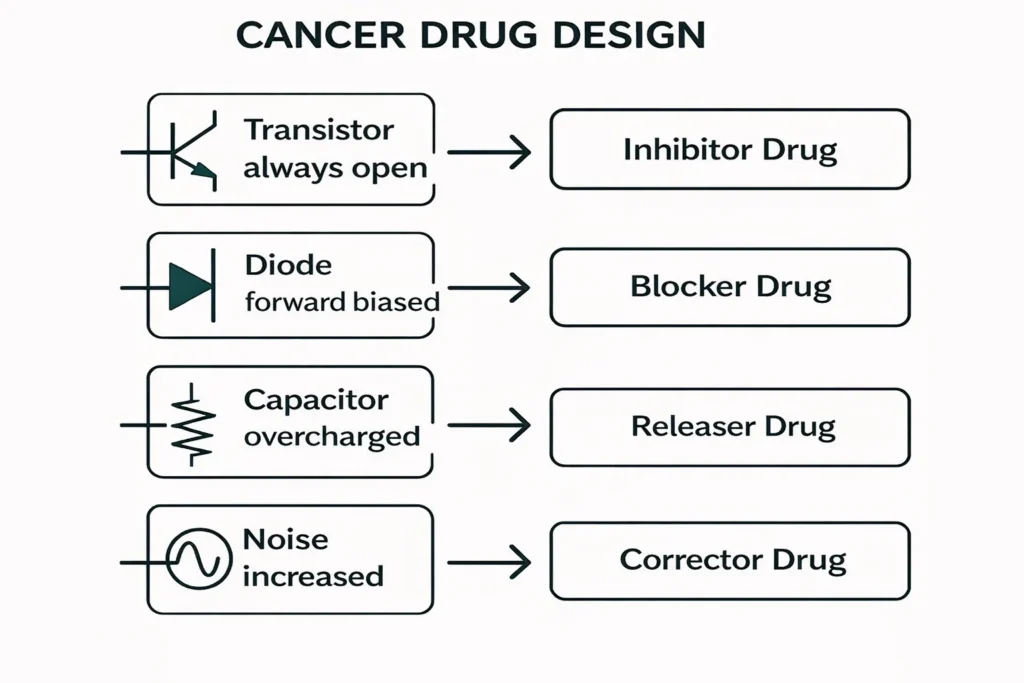
Drug design scheme according to circuit analogy
The following design answers the question, “Which remedy for which circuit failure?” Each block can be scored with adjustable parameters within the module.
1. Transistor modulators (on/off)
- Fault: Transistor continuously on — uncontrolled proliferation/signaling (e.g., oncogenic pathway).
- Drug rationale: Shut-off or partial modulator.
- Examples of targets:
- Kinase inhibitors: EGFR/ERBB, VEGFR, RAF/MEK.
- Nuclear receptor modulators: MR/GR/ER.
- Parameters: Gain (g_m) reduction, threshold (V_th) increase, saturation prevention.
2. Resistance modulators (ion channel/carrier)
- Fault: Resistance too low/high — excessive flow or blockage (ion homeostasis disorder).
- Drug logic: Enhancing (blocking) or decreasing (opening).
- Target examples:
- Ion channels: Na⁺, K⁺, Ca²⁺, Cl⁻ channels.
- Transporters: SGLT2, NHE3, ENaC, GLUT.
- Parameters: R_set, conductivity (1/R), noise (thermal).
3. Capacitor dischargers (energy/signal storage)
- Fault: Capacitor overload — signal/calcium/second messenger load.
- Drug logic: Discharge or store limiting.
- Target examples:
- Ca²⁺ modulation: SERCA inhibitor/activation, RyR modulation.
- mTOR/AMPK axis: Energy store/expenditure balance.
- Parameters: C_eff reduction, discharge rate (τ = R·C) adjustment.
4. Inductor boosters (energy reserve/flow stability)
- Fault: Inductor saturation — flow stability and reserve deficiency.
- Drug rationale: Reserve enhancer and stabilizer.
- Target examples:
- Mitochondrial function: ETC modulation, ROS reduction (Nrf2 concept).
- Osmotic gradient transporters: NKCC2, urea transporters.
- Parameters: L_eff enhancement, dI/dt limit smoothing.
5. Feedback control circuit (hormone/signal axes)
- Failure: Positive feedback lock-in — overamplification.
- Drug rationale: Adding negative feedback or limiting power.
- Target examples:
- RAAS/ADH/Endothelin, PI3K–AKT, MAPK.
- Parameters: Reducing loop gain (Aβ), increasing phase margin.
Variation module integration
Let’s design combinations by entering the following parameters between 0 and 1:
- Transistor gain distortion (T_gain): 0–1
- Ion conductivity deviation (Ion_cond): 0–1
- Cap load (Cap_load): 0–1
- Ind reserve weakness (Ind_reserve): 0–1
- Feedback overflow (FB_over): 0–1
Total treatment score:
TxScore= 𝑤𝑇 𝑇gain + 𝑤I Ioncond + 𝑤C Capload + 𝑤L Indreserve + 𝑤F FBover
Combination principles
- Arrangement:
1. Transistor modulation (multiplication control)
2. Feedback breakdown (network stability)
3. Resistor-capacitor regulation (current and storage)
4. Inductor reserve (durability)
- Risk monitoring: Electrolyte, blood pressure, metabolic load; avoid excessive damping in feedback interventions.
Notes
This design diagram is for educational purposes only and provides an idea at the general level of mechanism. Diagnosis and treatment require clinical decision-making; I cannot recommend personalized medication.
Chemical-atomic coupling through reverse logic
I map each block and fault type in the circuit analogy to chemical classes and atomic/electronic properties using reverse logic. The goal: to establish the chain: “circuit behavior → molecule types → atomic parameters”.
Circuit element → chemical class → atomic property mapping
| Circuit element | Malfunction/behavior | Chemical class (example target) | Atomic/electronic equivalent |
| Transistor (gain/gate) | Always on | Kinase inhibitors, receptor antagonists (EGFR, RAF/MEK, ER/AR) | Connectivity to d-orbital transition metals; π–π interacting aromatic rings; H-bond “gate” design. |
| Diode (directional) | Backflow/leakage | Adhesion and barrier stabilizers (nephrin/podocin modulators), integrin ligands. | Directional hydrogen bond networks with polar groups (–OH,–NH₂, –COOH); directionality with halogen bonding. |
| Resistance (conductivity) | Too low/high | Ion channel/carrier modulators (Na⁺/K⁺/Ca²⁺, SGLT2, ENaC) | Load/partial load distribution; σ/π donor–acceptor balance; coordination links. |
| Capacitor (storage unit) | Overfull | Second messenger and energy storage modulators (Ca²⁺, mTOR/AMPK, SERCA/RyR) | Chelating motifs (carboxylate, catechol); multiple coordination centers; high dielectric media preference. |
| Inductor (reserve/stability) | Saturation/reserve is low. | Mitochondrial function support, ROS modulators (Nrf2 pathway concept) | Redox-active rings (quinone, phenolic); transition metal interaction; electron delocalization |
| Noise (entropy) | Genetic instability | DNA repair/pathway modulators (PARP, HDAC, DNMT) | N-heteroaromatic rings; base stacking with π-stacking; polarization–hydrophobic equilibrium. |
| Feedback (loop gain) | Positive feedback lock | RAAS/ADH/Endothelin, PI3K–AKT, MAPK modulators | Adaptation to highly valuable binding sites; multiple pharmacophores; flexible conformer sequences. |
Atomic parameters and their effect on circuit behavior
- Electron delocalization: Aromatic/conjugated systems provide current-like flow; increase transistor-like gain.
- Coordination capability: Ligands that can bind with transition metals (Fe, Cu, Zn) regulate the “inductor/reserve” effect.
- Halogen and hydrogen bond orientation: Establish selective, unidirectional interaction networks that mimic diode orientation.
- Dielectric and polarity: In the capacitive storage analogy, solvent-molecule interactions determine charge storage (C_eff).
- Steric volume and conformation: In the resistivity analogy, this is the physical equivalent of channel passage/blocking.
Reverse mapping with pharmacophore motifs
- Shutter (transistor): Aromatic core + H-bond acceptor + hydrophobic pocket matching → kinase/GPCR shutdown.
- Inhibitor (diode): Directional polar arrangement (donor/acceptor pairs) + halogen bond → barrier leakage reduction.
- Depletor (capacitor): Chelating groups (EDTA-like, catechol) + Ca²⁺ dynamics modulation → reservoir depletion.
- Stabilizer (inductor): Redox-active rings + free radical buffer → reserve and flow stability.
- Noise regulator (entropy regulator): N-heteroaromatic base analogues + epigenetic modulation → noise (entropy) reduction.
Chemical design parameter panel (variation module compatible)
- Electronic structure:
- Aromatic delocalization: 0–1
- Polarity/dielectric interaction: 0–1
- Connectivity features:
- Halogen binding potential: 0–1
- H-bond donor/acceptor balance: 0–1
- Coordination (transition metal) ability: 0–1
- Geometry:
- Steric volume/cone barrier: 0–1
- Conformational flexibility: 0–1
By optimizing the chemical profile according to the “circuit failure” type using these parameters, we can generate a logical shortlist for target class selection.
Example reverse scenarios
- Transistor continuously on → gain reduction:
- Chemical profile: Aromatic kinase core, strong H-bond acceptor, hydrophobic pocket conformation.
- Atomic properties: High specificity with π–π stacking and d-orbital interaction.
- Diode leakage → directionality enhancement:
- Chemical profile: Small molecule containing halogen (Cl/Br) with a directional hydrogen bond motif.
- Atomic properties: Halogen bond (σ-hole) + polar strand, unidirectional bonding.
- Capacitor overcharged → Ca²⁺ discharge:
- Chemical profile: Polyfunctional chelator; combination with SERCA/RyR modulator.
- Atomic properties: Multiple coordination centers (O/N donor), high ligand domain strength.
- Inductor saturation → reserve increase:
- Chemical profile: Redox buffer (quinone/phenolic), mitokinetic support concept.
- Atomic properties: Electron accepting/donating capacity; controlled ROS interaction.
Conclusion
- Chemical-atomic reverse coupling in the circuit systematizes target class selection based on electronic structure, binding patterns, and geometric parameters.

