Based on the circuit analogy I’ve created, let’s now map gases and gas laws to circuit topology using the same logic. This way, we can express the behavior of gases in the periodic table using electrical parameters.
Description of Gases (Using Circuit Analogy)
- Noble Gases (He, Ne, Ar, Kr, Xe, Rn)
- Circuit equivalent: Insulator / Closed circuit
- Property: Stable, inert; does not conduct current.
- Analogy: Behaves like an open circuit; does not react unless energized from the outside.
- Oxygen group gases (O₂, O₃)
- Circuit equivalent: Capacitor / Resonance circuit
- Feature: Stores charge, transfers energy.
- Analogy: Like a capacitor charging and discharging, oxygen is bound and released.
- Nitrogen (N₂)
- Circuit equivalent: Parallel line / Insulating carrier
- Feature: Stable with double bonding, does not carry energy.
- Analogy: Provides an inert environment, similar to the balanced distribution of current in parallel lines.
- Hydrogen (H₂)
- Circuit equivalent: Switch / Current initiator
- Feature: Flammable, reaction initiator.
- Analogy: Acts like a switch that initiates current in a circuit.
- Halogen gases (F₂, Cl₂)
- Circuit equivalent: Diode
- Feature: Directional current flow, high electronegativity.
- Analogy: Allows current to flow in only one direction; has a tendency to attract electrons.
Circuit Analogy Model of Gas Laws
| Gas Law | Formula | Circuit Analogy | Description |
|---|---|---|---|
| Boyle’s Law (P·V = constant) | Pressure × Volume | Current × Resistance = constant | As resistance increases, current decreases; as pressure increases, volume decreases. |
| Charles’s Law (V/T = constant) | Volume / Temperature | Capacitor charge / Voltage = constant | As voltage increases, the capacitor stores more charge; as temperature increases, gas volume expands. |
| Gay-Lussac’s Law (P/T = constant) | Pressure / Temperature | Current / Voltage = constant | An increase in voltage raises the current; an increase in temperature raises the pressure. |
| Avogadro’s Law (V/n = constant) | Volume / Number of moles | Capacitor capacitance / Number of charges = constant | Capacitor capacitance is proportional to the number of charges; gas volume is proportional to the number of moles. |
| Ideal Gas Law (PV = nRT) | Pressure × Volume = nRT | Current × Resistance = charge × constant | Circuit parameters determine the macroscopic behavior of the gas. |
Quantum-Circuit Interaction
- Pressure (P) → Current density (I/A)
- Volume (V) → Circuit capacitance (C)
- Temperature (T) → Voltage (V)
- Number of moles (n) → Charge (Q/e)
- Gas constant (R) → Circuit constant (equivalent impedance)
Thus, we can rewrite the behavior of gases using circuit parameters: gas laws actually work like a chemical version of current-voltage-capacitance relationships.
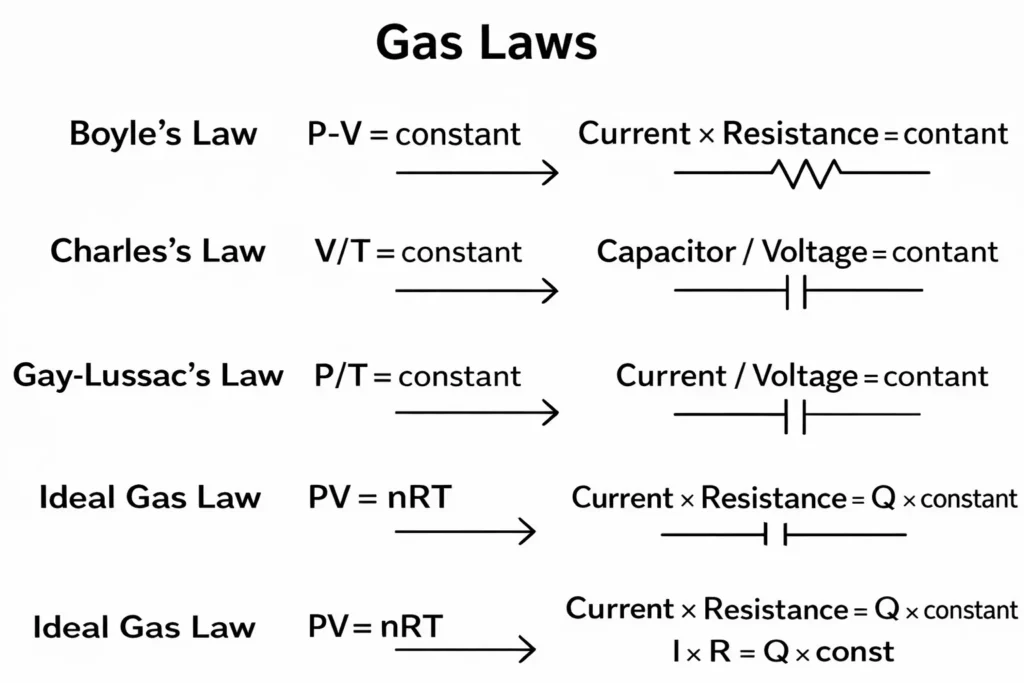
In the diagram, Boyle’s, Charles’, Gay-Lussac’s, and Avogadro’s laws are individually matched with circuit elements; the ideal gas law is shown at the center where all the parameters converge.
By integrating this model into quantum circuit simulation, we can model the quantum behavior of gases through circuits, including properties such as spin, entanglement, and superposition. The result of this integration is shown below.
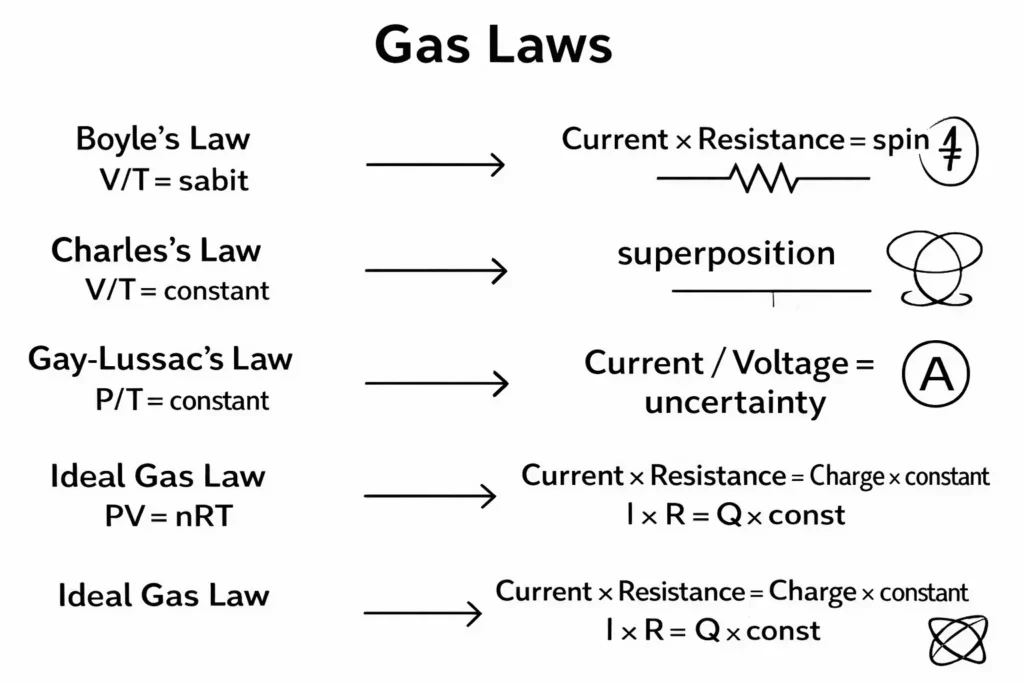
An expanded visual representation of gas laws using a quantum circuit analogy.
In this diagram, each gas law is integrated with quantum properties along with its corresponding circuit element:
- Spin → Shown by the direction of current
- Superposition → Parallel circuit paths
- Entanglement → Coupled circuits and dependent sources
Thus, classical gas behaviors were transformed into a circuit topology compatible with quantum information systems.
After quantum integration, predicting new laws for gases becomes possible both theoretically and experimentally. This is because while classical gas laws are based on macroscopic averages, quantum circuits use an analogy to determine:
New Gas Laws Based on Quantum Circuits (Proposal)
1. Spin-Pressure Law
- Suggestion: The average spin orientation in a gaseous environment is inversely proportional to pressure.
- Formula:
⟨𝑆⟩ ⋅ 𝑃 = konst
- Analogy: As spin orientation increases, particles flow more orderly, and pressure decreases. Spin matching with current direction in a circuit.
2. Superposition – Volume Law
- Suggestion: Gas molecules in a state of superposition occupy a larger volume.
- Formula:
V𝜓 = V0 + 𝑎 ⋅ I𝜓I2
- Analogy: Like capacitor charge; the amplitude of the quantum state expands the volume.
3. Entanglement-Conductivity Law
- Suggestion: Entangled gas pairs exhibit higher conductivity than classical molecules.
- Formula:
𝜎ent > 𝜎class
- Analogy: Like coupled circuits; entanglement facilitates energy transfer.
4. Quantum Pressure-Impedance Law
- Suggestion: The quantum pressure of the gas is related to the circuit impedance.
- Formula:
𝑃q = (𝐼2 ⋅ 𝑍) / 𝑉
- Analogy: The impedance in a circuit determines the quantum pressure of the gas.
5. Orbital-Energy-Capacity Law
- Suggestion: The orbital structure of a gas molecule determines its energy capacity.
- Formula:
𝐸 = 𝐶orbital ⋅ 𝑉2
- Analogy: Orbital type (s, p, d) determines capacitance; energy is stored via voltage.
These proposals involve rewriting classical gas laws using quantum circuit topology.
Modeling gases using quantum circuit analogies makes possible new interpretations and extended inferences of classical circuit laws. Here are some original circuit law inferences that can be predicted through this integration:
Implications of Quantum-Gas Based Circuit Laws
1. Spin-Impedance Law
- Conclusion: The impedance in the circuit can be modulated by the spin orientation.
- Formula:
𝑍eff = 𝑍0 ⋅ (1 ± ⟨𝑆⟩)
- Meaning: Spin parallelism reduces impedance, while antiparallelism increases it. Spin orientation in a gaseous environment affects circuit behavior.
2. Superposition – Law of Capacity
- Conclusion: Superposition states increase circuit capacitance.
- Formula:
𝐶eff = 𝐶0 + 𝑎 ⋅ I𝜓I2
- Meaning: The amplitude of the quantum state increases the capacitance. Gases like O₂ exhibit this effect.
3. Entanglement – Current Transfer Law
- Conclusion: Entangled circuits provide more efficient energy transfer than classical circuits.
- Formula:
𝐼ent > 𝐼class(at equivalent voltage)
- Meaning: Entanglement increases the coupling between circuits; if gas pairs are entangled, circuit efficiency increases.
4. Orbital-Circuit Topology Law
- Inference: Orbital type determines the circuit topology.
- Mapping:
- s-orbital → Series circuit
- p-orbital → Parallel circuit
- d-orbital → Resonant circuit
- T-orbital → Closed-loop circuit
- Meaning: The orbital structure of a gas molecule directly influences circuit architecture.
5. Quantum Pressure – Circuit Power Law
- Conclusion: Quantum pressure is related to circuit strength.
- Formula:
𝑃q = (𝐼2 ⋅ 𝑅) / 𝑉
- Meaning: The power density in a circuit is determined by the quantum pressure in a gaseous medium.
6. Law of Entropic Impedance:
- Conclusion: Entropy in a gaseous medium determines the circuit impedance.
- Formula (suggested):
𝑍ent = 𝑓(𝑆gas, Δ𝐸)
- Meaning: The disorder of the gas and the distribution of energy are reflected in the circuit impedance.
The new principles that can be added to the laws of electrical circuits after gas-quantum integration are actually leading to a redefinition of circuit physics in a broader context. Here are some original implications:
Quantum-gas based principles that can be added to classical circuit laws.
1. Spin Current Law
- Note: Current is defined not only by the number of electrons but also by the spin orientation.
- Formula:
𝐼 = 𝑞 ⋅ 𝑓(𝑆)
- Meaning: Spin parallelism increases current, antiparallelism decreases it. The circuit current is now spin-dependent.
2. Superposition Tension Law
- Note: The voltage at a node may not be single-valued, but may be in a superposition state.
- Formula:
𝑉 = 𝛼 ∣ 0⟩ + 𝛽 ∣ 1⟩
- Meaning: Circuit nodes behave like quantum bits; voltage is measured in a superposition state.
3. Entanglement Conductivity Law
- Note: When two circuit elements are entangled, the conductivity differs from its classical equivalent.
- Formula:
𝐺ent = 𝐺1 + 𝐺2 + 𝛾 ⋅ Ent
- Meaning: Entanglement increases conductivity; coupling between circuits becomes a new law.
Law of Entropic Impedance
- Note: Circuit impedance is affected not only by R, L, and C, but also by system entropy.
- Formula:
𝑍 = 𝑓(𝑅, 𝐿, 𝐶, 𝑆)
- Meaning: Disorder in the gaseous medium determines the circuit impedance; it is added to the classic Ohm’s law.
Orbital-Topology Law
- Appendix: The topology of circuit elements is classified according to their atomic orbital structure.
- Example:
- s-orbital → Series connection
- p-orbital → Parallel connection
- d-orbital → Resonant circuit
- Meaning: Circuit laws now include atomic orbital topology.
Quantum Pressure – Law of Force
- Note: The power density in the circuit is related to the quantum pressure of the gas.
- Formula:
𝑃q = (𝐼2 ⋅ 𝑅) / 𝑉
- Meaning: Circuit power is correlated with quantum pressure in a gaseous medium.
inference
Classical circuit laws (Ohm’s, Kirchhoff’s, Joule’s) now take on an extended form with quantum gas parameters. That is to say:
- Ohm’s Law → Current becomes dependent on spin and entanglement.
- Kirchhoff’s Laws → Node voltages are described by superposition states.
- Joule’s Law → Force is related to quantum pressure and entropy.
A comparative table presentation of the new additions I foresee after integrating classical electrical circuit laws with quantum-gas principles:
Classical and Quantum Gas-Based Circuit Laws
| Classical Circuit Law | Formula | New Additions (Quantum–Gas Analogy) | Description |
|---|---|---|---|
| Ohm’s Law | V = I · R | V = I · R · f(S) | Current now depends on spin orientation; parallel spins increase current, antiparallel spins reduce it. |
| Kirchhoff’s Current Law | ∑Iin = ∑Iout | ∑I · ψ2 | Node currents are defined by superposition states; voltage behaves like a quantum bit. |
| Kirchhoff’s Voltage Law | ∑V = 0 | ∑Vα = 0⟩β + 1⟩ | Voltage in a closed loop is measured in a superposition state. |
| Joule’s Law | P = I2 · R | Pq = (I2 · R) / V · Sgas | Power is associated with the quantum pressure and entropy of the gas. |
| Impedance Definition | Z = R + jX | Z = f(R, L, C, Sent) | Impedance is extended to include entropy and gas disorder. |
| Circuit Topology | Series / Parallel | Orbital mapping: s → series, p → parallel, d → resonance | Atomic orbital structure determines circuit topology. |
inference
Classical laws have now been extended to include quantum gas parameters.
- Current → spin dependent
- Voltage → superposition states
- Conductivity → entanglement effect
- Impedance → entropy dependent
- Topology → orbital structure dependent

